|
So, this makes the ion have a negative charge. When we gain more electrons, we can know that the amount of electrons is higher than the proton. So, we can conclude that this ion has a positive charge and vice versa. So, the dominant charge of the certain ion is the proton. If the ion loses the electron, this means that the negative charge is lesser than the positive charge (proton). PERIODIC TABLE OF THE ELEMENTS ©2019 Lanthanoids Actinoids 1 K 2 2 2 L 8 2 6 3 M 18 2 6 10 4 N 32 2 6 10 14 5 O 32 2 6 10 14 6 P 18 2 6 10 7 Q 8 2 6 8 R 2 2 Energy Level ELECTRON SHELLS Shell Name Max. Actually, to understand the ion, we can see how the atoms lose their negative charge (electron). The Natural formation of ions is: Group 1 elements form +1 ions. Actually, we have discussed this before above. This site provides thermochemical, thermophysical, and ion energetics data compiled by NIST under the Standard Reference Data Program. As you can see from the partial table shown above the Groups of the periodic table each form a unique charge of ion. shows the charge of the metal ions in that compound. When we download a periodic table of ions, we also need to know about how to determine an ion. The number in the name of the compound close compound A substance formed by the chemical union of two or more elements. The number of electrons is not included in the. This periodic table contains all 118 elements along with each element’s atomic number, symbol, name, atomic mass, electron shell configuration, and most common charges (oxidation states). For example, 146 C and 126 C are the nuclear symbols for the isotopes carbon-14 and carbon-12, respectively. This periodic table is a periodic table with charges PDF optimized to fit on a single sheet of paper. Where: A is the mass number or number of nucleons (protons + neutrons) Z is the atomic number or number of protons. Then, if it is reversed, which means the amount of electrons is higher than the proton, we can call it anion. The general form for the notation is: AZ X. The cation is where the positive charge (proton) has more amount than the electron. SODIUM Na+ MAGNESIUM Mg2+ When there is more than one common ion, the more common ion is shown on top. Note that CuO has only one copper ion, while Cu 2 O has two copper ions. 
Based on position on the periodic table, the oxide ion has a charge of 2. 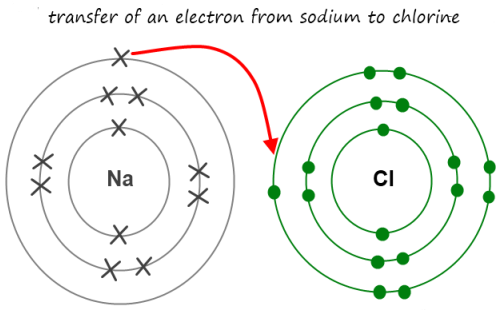
The first two groups have elements with only one or two electrons in their outer shells, groups 1 ns 1 and 2 ns 2 respectively. Ion formula Elements for which no ion is shown are those which do not commonly form ions, or superheavy elements whose chemistry is currently unknown. Write the name leaving room for a Roman numeral, since copper forms ions with variable charges. There are two kinds of ions in the periodic table of ions. The position of an element on the periodic table tells about the type of ions it will form, with the elements on the right forming anions (negative charge) and those further to the left forming cations (positive charge). However, it is pretty normal to be found that there is an imbalance of the charge, whether the number of protons is higher or the number of electrons is higher. Well, there are two kinds of ions on the periodic table.Īctually, if an atom or atoms that have equal numbers of negative charge (electrons) and positive charge (proton), then they can be defined as a neutral atom. Yes, one of the things that is required to be understood in the beginning is the ions themselves. Yes, we can call this a periodic table of ions.īefore we download the periodic table of irons, we actually need to understand the basic things inside the periodic table of ions. (Remember from our conventions for writing formulas that we do not write a 1 subscript if there is only one atom of a particular element present.When it comes to learning Chemistry lessons, we may have seen our teacher introduce us with a sheet of the grid that consists of many abbreviations as a symbol of the elements. The formula Na 2Cl 2 also has balanced charges, but the convention is to use the lowest ratio of ions, which would be one of each.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
